d) There is a definite drawback to running with one reactant in excess rather than feeding the reactants in stoichiometric proportion. 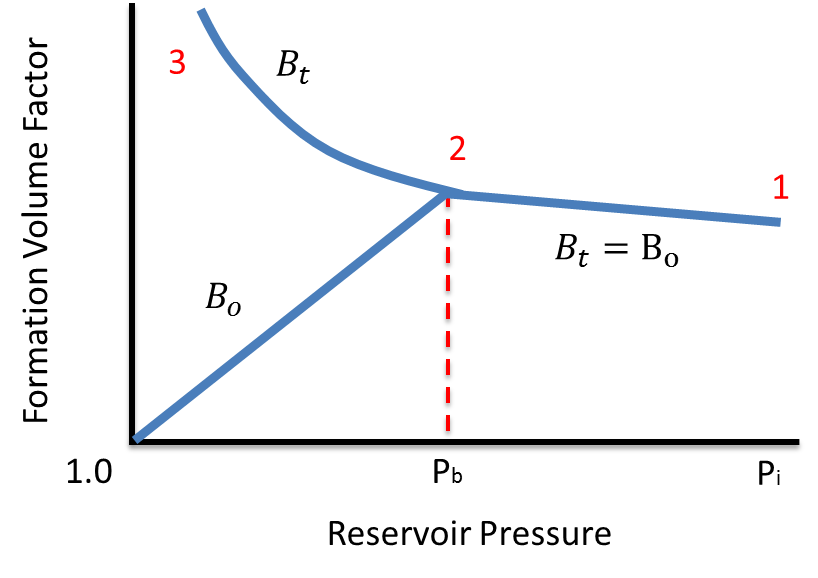
Operator: Your next question comes from the line of Michael Lasser with UBS. 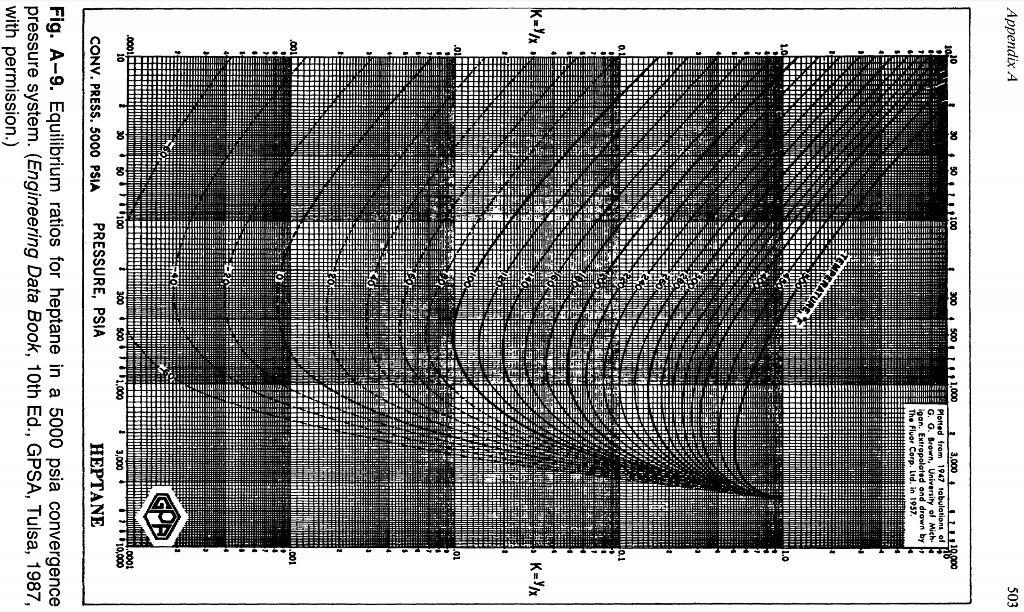
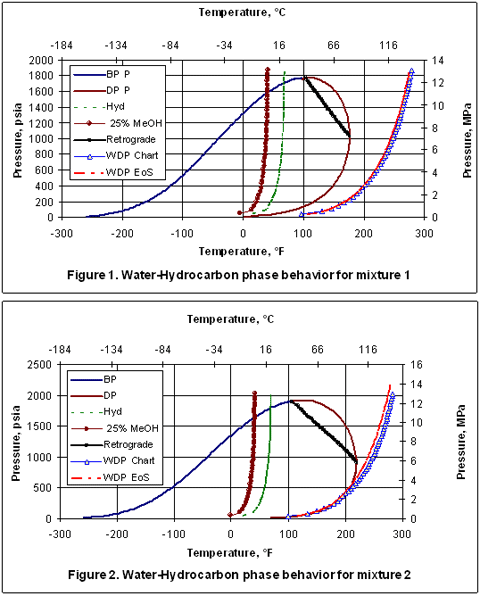
Bubble point pressure was deduced from the raw differential pressure transducer (DPT) measurement from the sense line shown in Fig. P ∗ = 1 0 A − B T C \mathrm 4 × 1 0 6 metric tons of ethane per year, assuming that the reaction goes to completion and that the process operates for 24 hours a day, 300 days a year. Bubble point pressure and gas-oil ratio data are obtained both by direct measurement and by calculation. as critical temperature, critical pressure and acentric factor, to calculate. Antoine equation is used for determine the vapor pressure: Phase equilibrium properties, such as bubble-point, hydrocarbon dewpoint.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
